Figure 3. Decontamination methods. a Gauze soaked in saline applied using a sawing motion. b Ultrasonic scaler (SUPRASSON P-MAX, Satelec-Acteon group, Bordeaux, France, power setting: P5, tip: Implant Protect IP3L/R).
Figure 3. Decontamination methods
author: Motohiro Otsuki, Masahiro Wada, Masaya Yamaguchi, Shigetada Kawabata, Yoshinobu Maeda Kazunori Ikebe | publisher: drg. Andreas Tjandra, Sp. Perio, FISID
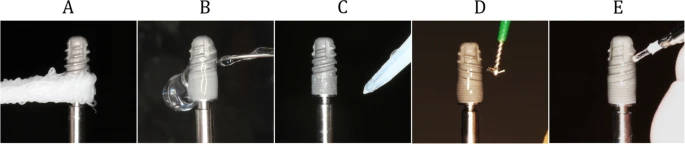
Figure 3. Decontamination methods. a Gauze soaked in saline applied using a sawing motion. b Ultrasonic scaler (SUPRASSON P-MAX, Satelec-Acteon group, Bordeaux, France, power setting: P5, tip: Implant Protect IP3L/R). c Air abrasives (AIR-FLOW MASTER PIEZON®, EMS, Nyon, Switzerland, power setting: water flow 100%, air pressure 75%, powder: AIR-FLOW® PERIO POWDER, nozzle: PERIO-FLOW® nozzles, distance from the nozzle to the implant 2 mm). d Rotary stainless steel instrument (iBrush, NeoBiotech©, Los Angeles, USA, rotating speed 1500 rpm). e Er:YAG laser (Erwin AdvErL, J.Morita©, Kyoto, Japan, power setting 60 mJ/pulse, 10 pps, tip: C600F, distance from the tip to the implant 2 mm)
Serial posts:
-
Evaluation of decontamination methods of oral biofilms formed on screw-shaped, rough and machined surface implants: an ex vivo study
-
Background : Evaluation of decontamination methods of oral biofilms formed on screw-shaped, rough and machined surface implants
-
Materials & methods : Evaluation of decontamination methods on implants (1)
-
Materials & methods : Evaluation of decontamination methods on implants (2)
-
Materials & methods : Evaluation of decontamination methods on implants (3)
-
Results : Evaluation of decontamination methods on implants (3)
-
Discussion : Evaluation of decontamination methods on implants (1)
-
Discussion : Evaluation of decontamination methods on implants (2)
-
Discussion : Evaluation of decontamination methods on implants (3)
-
Discussion : Evaluation of decontamination methods on implants (4)
-
Discussion : Evaluation of decontamination methods on implants (5)
-
Discussion : Evaluation of decontamination methods on implants (6)
-
Discussion : Evaluation of decontamination methods on implants (7)
-
Discussion : Evaluation of decontamination methods on implants (8)
-
Discussion : Evaluation of decontamination methods on implants (9)
-
Figure 1. Hard resin splint model carrying 6 implants
-
Figure 2. GC Aadva® implant; 3.3-mm diameter, 8-mm length
-
Figure 3. Decontamination methods
-
Figure 4. SEM analysis of 4 areas. 1 Rough surface—microthread area
-
Figure 5. Quantitative analysis of CFU counts on implants
-
Figure 6. Comparison of cleansability of each decontamination method
-
Table 1 Qualitative evaluation by SEM analysis of micro- and macrothread areas of rough surface implants
-
Table 2 Qualitative evaluation by SEM analysis of micro- and macrothread areas of machined surface implants
-
Table 3 Quantitative analysis of CFU counts

- Evaluation of decontamination methods of oral biofilms formed on screw-shaped, rough and machined surface implants: an ex vivo study
- Background : Evaluation of decontamination methods of oral biofilms formed on screw-shaped, rough and machined surface implants
- Materials & methods : Evaluation of decontamination methods on implants (1)
- Materials & methods : Evaluation of decontamination methods on implants (2)
- Materials & methods : Evaluation of decontamination methods on implants (3)
- Results : Evaluation of decontamination methods on implants (3)
- Discussion : Evaluation of decontamination methods on implants (1)
- Discussion : Evaluation of decontamination methods on implants (2)
- Discussion : Evaluation of decontamination methods on implants (3)
- Discussion : Evaluation of decontamination methods on implants (4)
- Discussion : Evaluation of decontamination methods on implants (5)
- Discussion : Evaluation of decontamination methods on implants (6)
- Discussion : Evaluation of decontamination methods on implants (7)
- Discussion : Evaluation of decontamination methods on implants (8)
- Discussion : Evaluation of decontamination methods on implants (9)
- Figure 1. Hard resin splint model carrying 6 implants
- Figure 2. GC Aadva® implant; 3.3-mm diameter, 8-mm length
- Figure 3. Decontamination methods
- Figure 4. SEM analysis of 4 areas. 1 Rough surface—microthread area
- Figure 5. Quantitative analysis of CFU counts on implants
- Figure 6. Comparison of cleansability of each decontamination method
- Table 1 Qualitative evaluation by SEM analysis of micro- and macrothread areas of rough surface implants
- Table 2 Qualitative evaluation by SEM analysis of micro- and macrothread areas of machined surface implants
- Table 3 Quantitative analysis of CFU counts